Our client, a US pharmaceutical company managing an active portfolio of Phase II–IV clinical trials with offices and research centers across Europe, needed to unify fragmented trial operations spanning EDC (Electronic Data Capture), CTMS (Clinical Trial Management System), Excel, safety systems, and local databases.
Computools used platform engineering, systems integration, and workflow automation to develop a centralized operational layer that connected existing systems, standardized data flows, and improved visibility across studies. This platform reduced manual coordination, enhanced audit readiness, and established a scalable foundation for future digital growth in clinical operations.
The company manages multiple parallel clinical studies and works with contract research organizations (CROs), study coordinators, data managers, and regulatory teams across a complex research environment.
As the number of active trials grew, maintaining operational consistency became challenging. Study data, site information, and reporting workflows were spread across multiple systems, making it difficult to monitor progress, coordinate teams, and ensure compliance.
To support sustainable growth, the company needed a more structured operating model that could centralize trial oversight, reduce manual effort, and improve control across study execution.
Fragmented systems and limited process visibility constrained the company’s clinical trial operations. Each study used disconnected tools such as EDC, CTMS, Excel, safety systems, and local databases. Limited integrations failed to support all critical workflows. Teams manually duplicated data, reconciled inconsistencies, and spent considerable time searching for operational information.
This fragmentation slowed recruitment tracking, delayed site activation, complicated reporting, and made audit preparation highly resource-intensive. Data accuracy depended too heavily on manual coordination, while the lack of a single operational view made it difficult to assess study status in real time.
As study volume increased and regulatory expectations tightened, the existing setup raised the risk of non-compliance, higher costs, and delays in bringing new therapies to market. Without a unified platform, the company’s ability to scale clinical operations efficiently remained limited.
Computools developed and deployed a unified clinical trials platform, ClinUnity, adding a centralized operational layer to the client’s existing systems.
The solution integrated EDC, CTMS, safety systems, spreadsheets, and internal data sources into a coordinated environment with a unified data model and automated information flows. Standardized workflows minimized manual handoffs and improved consistency in study setup, site management, recruitment tracking, data review, and compliance activities.
The ClinUnity platform features a unified trial dashboard, site, and patient tracking, automated data ingestion, workflow orchestration, role-based access control, and comprehensive audit trails. It provides near-real-time operational visibility, fast search, secure data handling, and full traceability of critical actions.
The modular architecture provides a stable foundation for scaling to additional studies and future integration of predictive analytics.
The ClinUnity platform significantly improved operational efficiency and trial coordination.
The platform now serves as a core component of the client’s clinical operations infrastructure, reducing coordination overhead and providing a stronger foundation for scaling future R&D and trial management initiatives.
Computools was chosen for its expertise in healthcare software engineering, data integration, and secure platform delivery within regulated environments.
The project required a unified operational model for fragmented systems, reliable management of sensitive clinical data, and a scalable architecture that would not disrupt existing tools.
The team delivered:
All engineering decisions focused on reducing operational friction, improving traceability, and supporting more controlled study execution.
The client operated in a complex clinical research environment where multiple studies were managed in parallel through disconnected systems and manual coordination. While core applications supported specific functions, they did not provide a unified operational layer across study setup, site management, recruitment tracking, and reporting.
As the number of active trials grew, the limitations of this approach became clear. Teams relied on Excel, local tracking files, and manual updates between systems. This led to duplicated, delayed, or inconsistent information, making oversight across studies difficult.
Meanwhile, the company faced growing pressure to accelerate timelines, maintain audit readiness, and reduce the operational cost of trial execution. To support future growth, it required a platform to centralize control without replacing its existing clinical systems.
The goal was not simply to deploy new software but to create a single operational environment across fragmented processes. The first priority was to build a centralized platform layer that could connect fragmented systems and standardize operational workflows across multiple studies. This included aligning study, site, patient, and compliance-related data into a consistent structure that could support fast access, reliable reporting, and coordinated execution.
The architecture was designed as a scalable platform for multi-study clinical trial management, with strong emphasis on secure integrations, workflow traceability, and role-specific access to operational information.
Computools served as the end-to-end technology partner, transforming fragmented clinical operations into a structured, scalable, and compliant digital environment.
The team led:
By aligning platform logic with actual clinical workflows, Computools ensured the solution supported daily study execution instead of introducing another disconnected layer.
A key architectural decision was to implement a unified data model that standardized information across studies and systems. This reduced inconsistency, simplified reporting, and provided teams with a more reliable operational view.
Another important decision was to adopt an event-driven architecture to enable timely updates and coordinated workflows across connected systems. This improved responsiveness without requiring the client to replace existing tools.
The modular structure allowed phased platform deployment, beginning with core operational capabilities and expanding to additional studies over time. This reduced implementation risk, accelerated adoption, and provided a practical path for broader digital transformation in clinical operations.
For the next phase, the client is considering predictive analytics to forecast patient dropout risk and anticipate study delays. This would extend the platform from centralized coordination to more proactive operational planning.
The platform was designed to transform fragmented clinical operations into a clear, structured, and decision-ready user experience. It aims to support operational teams managing multiple studies, systems, and compliance-sensitive workflows without adding complexity.
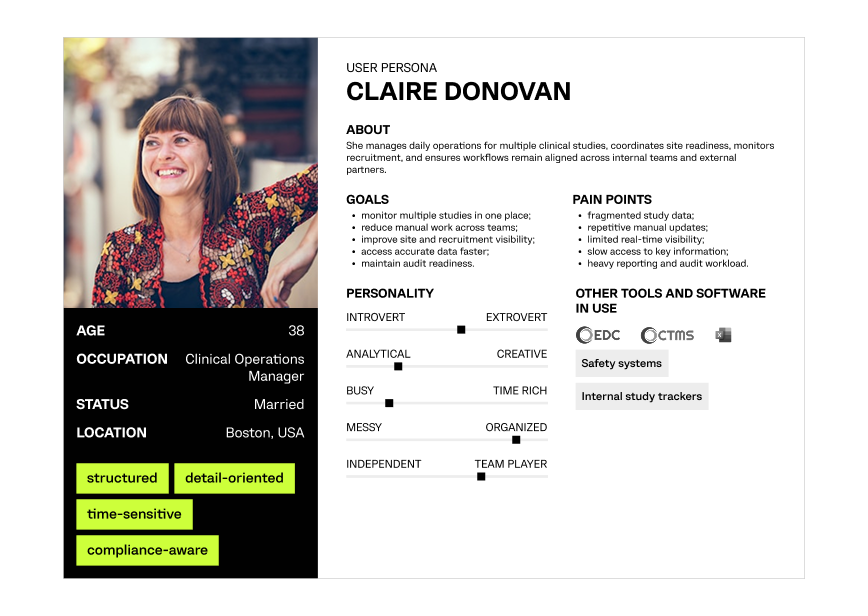
Creating a detailed profile of the primary operational user to align the platform with real multi-study clinical workflows.
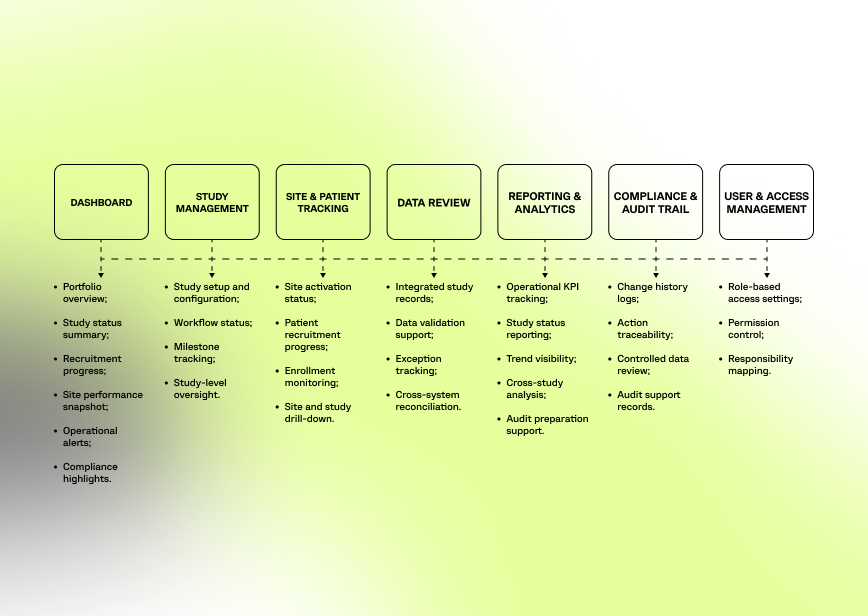
The platform structure was designed to mirror the operational flow of multi-study clinical trial management and bring related workflows into one connected environment.
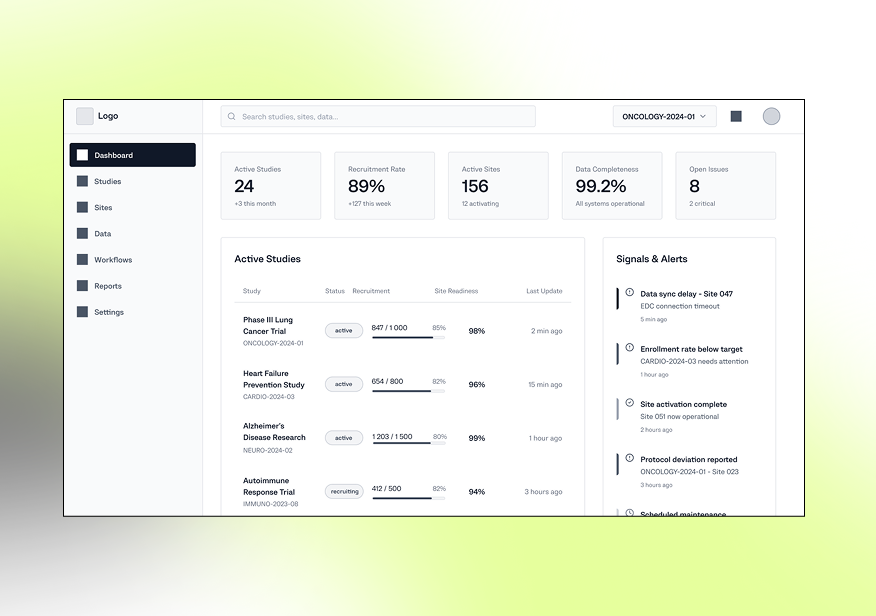
Low-fidelity layouts focused on fast navigation across study data, clear operational visibility, traceable workflows, and minimal friction between monitoring, validation, reporting, and compliance tasks.
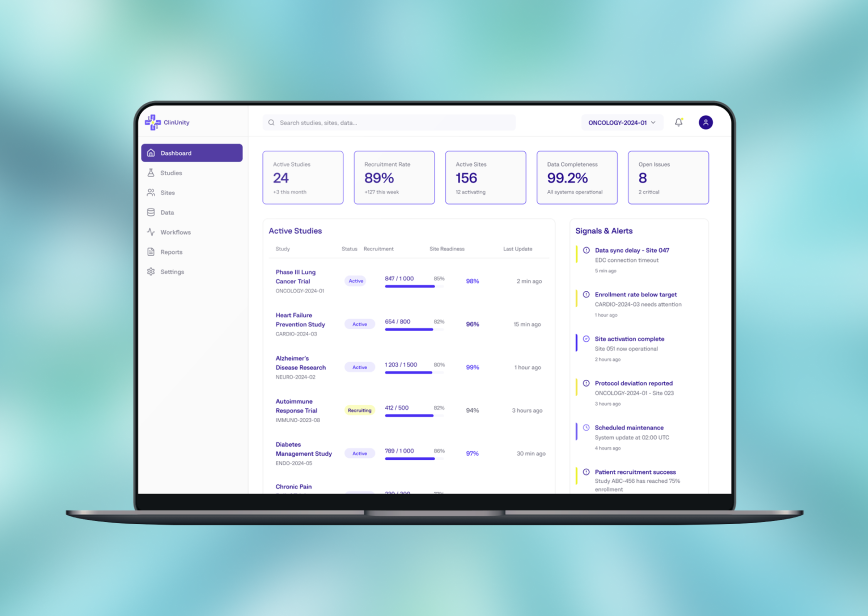
The final interface delivers centralized dashboards, structured study views, operational alerts, and workflow-oriented navigation designed for fast execution in a compliance-sensitive clinical environment.

PYTHON
Python is used as the core backend language for data processing, integration logic, and workflow automation. It is well-suited for building scalable services that handle fragmented clinical trial data and support future analytical extensions.

FASTAPI
FastAPI is used to build high-performance APIs for integrating EDC, CTMS, safety systems, spreadsheets, and internal data sources. It enables clean service architecture and efficient communication between platform modules.

REACT
React powers the platform’s web interface, including dashboards, study views, site tracking, reporting, and user management. It enables responsive and role-specific user experiences for clinical operations, data managers, and compliance teams.

AWS CLOUD
AWS Cloud provides secure, scalable infrastructure for hosting platform services, APIs, storage, and monitoring components. It supports enterprise-grade availability, security controls, and long-term platform growth.
DOCKER
Docker is used to containerize platform services and deployment environments. It ensures consistency across development, testing, and production while simplifying maintenance and release management.

POSTGRESQL
PostgreSQL is used as the main structured data store for studies, sites, workflows, users, and audit-related records. It provides transactional consistency and reliability for operational processes in a regulated environment.

APACHE KAFKA
Apache Kafka supports event-driven communication across services and connected systems. It keeps operational data in sync, improves responsiveness, and supports the scalable handling of study-related updates.

KEYCLOAK
Keycloak is used for authentication, authorization, and role-based access control. It strengthens user governance and helps protect sensitive clinical workflows through secure access management.
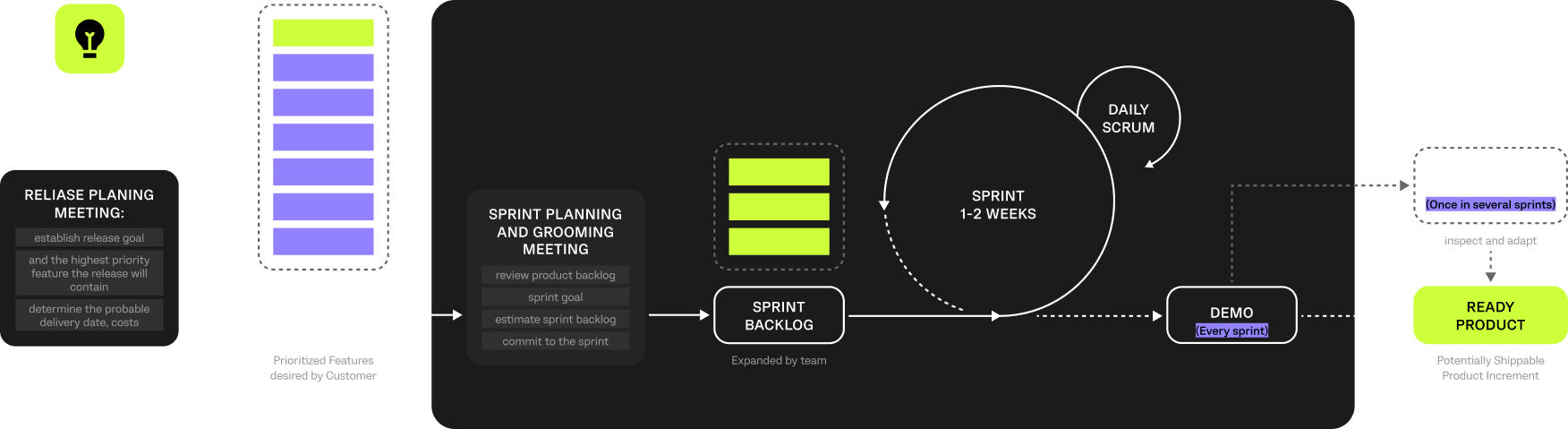
The project was managed using Agile with Scrum to enable iterative delivery in a complex, compliance-sensitive clinical environment.
Regular sprint cycles, reviews, and stakeholder feedback ensured the platform aligned with operational workflows, integration priorities, and regulatory requirements. This approach reduced delivery risks, supported phased implementation, and allowed the solution to adapt to the client’s evolving clinical operations.
We used to spend too much time moving between systems, checking spreadsheets, and piecing together the current status of each study. With the new platform, our teams have a much clearer view of operations, and a lot of the routine manual work is gone. It has made coordination faster, reporting easier, and audit preparation far less stressful.